When modeling the transport of contaminants through soil and groundwater, not all pollutants are conservative. Many substances, such as radionuclides or biodegradable organic compounds, decrease in concentration over time due to internal decay processes.
In GAEA Technologies POLLUTEv8, accounting for this loss is critical for accurate long-term impact assessments. Here is a step-by-step guide on how to implement decay in your models.
Understanding the Decay Constant
POLLUTEv8 uses a first-order decay model. The rate of mass loss is governed by the decay constant (). The relationship between the concentration (C) and time (t) is expressed as:
Where:
- C0 is the initial concentration.
- is the decay constant (units of
1/time).
Step 1: Calculate Your Decay Constant
Before entering data into the software, you must convert your contaminant’s half-life (t/1/2) into a decay constant. Use the following formula:
Step 2: Inputting Decay Data in POLLUTEv8
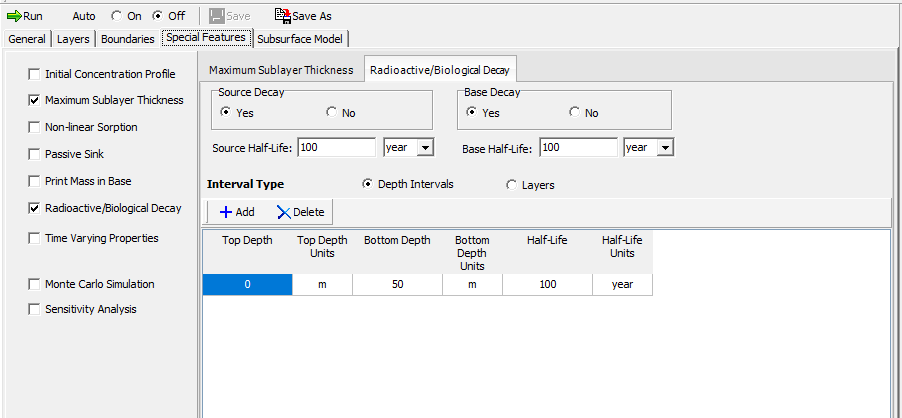
Radioactive and biological decay are entered on the Special Features tab. The half-lives are specified for each depth interval experiencing decay. In addition to the layers, decay can also be specified at the source and base of the model.
Typical Values of Radioactive and Biological Decay
In contaminant transport modeling, decay constants vary significantly depending on whether you are dealing with radioactive isotopes (which have fixed physical half-lives) or biological contaminants (which depend on environmental conditions like oxygen and temperature).
Below are typical values you might use in POLLUTEv8, categorized by contaminant type.
Radioactive Decay Constants
Radioactive decay is predictable and follows a strict physical half-life. These values are constant regardless of the soil medium.
| Radionuclide | Typical Half-life |
|---|---|
| Tritium | 12.32 years |
| Strontium-90 | 28.8 years |
| Cesium-137 | 30.17 years |
| Plutonium-239 | 24,110 years |
| Carbon-14 | 5,730 years |
2. Biological Decay Constants (Biodegradation)
Unlike radionuclides, biological decay constants for organic compounds (like petroleum hydrocarbons or chlorinated solvents) are highly variable. They are often reported as a range because “site-specific” conditions (pH, moisture, microbes) matter.
| Compound | Typical Half-life Range |
|---|---|
| Benzene | 10 – 700 days |
| Toluene | 2 – 100 days |
| Vinyl Chloride | 70 – 1,100 days |
| DCE (Dichloroethene) | 100 – 1,000 days |
Summary
Modeling decay in POLLUTEv8 requires converting a substance’s half-life into a first-order decay constant (). This value is then applied to the transport properties of the individual soil layers and, if applicable, the source boundary condition. This ensures that the model reflects the natural reduction of contaminant mass over the simulation period, preventing overly conservative (and potentially expensive) engineering designs.