Introduction
Predicting how contaminants migrate through soil and groundwater systems is one of the central challenges in hydrogeology and environmental engineering. When pollutants are released into the subsurface environment—from industrial spills, landfill leachate, agricultural chemicals, or mining activities—their movement is controlled by a complex combination of physical and chemical processes. These processes include advection, dispersion, diffusion, chemical reactions, and sorption.
Among these mechanisms, sorption plays a critical role because it controls how contaminants interact with soil and rock surfaces. Sorption can significantly slow contaminant migration by transferring pollutants from the aqueous phase (groundwater) to the solid phase (soil or mineral surfaces). This interaction effectively reduces the amount of contaminant moving with groundwater flow.
Many groundwater transport models simplify sorption behavior using linear sorption models, which assume that the amount of contaminant sorbed onto soil particles is directly proportional to the contaminant concentration in water. While this assumption is convenient and widely used, it does not always reflect real environmental conditions.
In many natural systems, sorption behavior is non-linear, meaning that the relationship between dissolved concentration and sorbed concentration changes with contaminant levels. Non-linear sorption occurs because sorption sites on soil particles may become saturated, chemical reactions may vary with concentration, or different sorption mechanisms may dominate under different environmental conditions.
Accurately representing non-linear sorption processes is essential for predicting contaminant plume migration, evaluating environmental risks, and designing remediation strategies. This article explores the concept of non-linear sorption in contaminant transport modeling, explains the mathematical models used to represent it, and discusses its implications for groundwater contamination studies.
Sorption Processes in Subsurface Environments
Sorption refers to the interaction between dissolved contaminants and solid materials in soil or rock formations. It includes two main mechanisms: adsorption and absorption.
Adsorption
Adsorption occurs when contaminants attach to the surface of mineral particles or organic matter. Soil minerals such as clays often have large surface areas and charged surfaces that attract dissolved ions and molecules.
Factors influencing adsorption include:
- mineral composition
- surface charge properties
- pH of groundwater
- ionic strength
- contaminant chemistry
Clay minerals and iron oxides typically exhibit strong adsorption properties.
Absorption
Absorption occurs when contaminants dissolve into the internal structure of solid materials, such as organic matter in soils.
Hydrophobic organic contaminants often partition into organic carbon in soils and sediments, reducing their mobility in groundwater.
Linear Sorption Models
In many environmental models, sorption is represented using a linear distribution coefficient (Kd).
The linear sorption relationship can be expressed as:
S = K_d C
Where:
- S = concentration of contaminant sorbed onto soil
- Kd = distribution coefficient
- C = dissolved contaminant concentration
This equation assumes that sorption sites are unlimited and that the ratio between sorbed and dissolved concentrations remains constant.
While linear sorption models are simple and computationally efficient, they often fail to capture complex sorption behavior observed in laboratory experiments and field studies.
Why Sorption Can Be Non-Linear
In many environmental systems, sorption behavior deviates from the linear assumption.
Non-linear sorption occurs when the relationship between dissolved concentration and sorbed concentration is not proportional.
Several factors can cause non-linear sorption.
Limited Sorption Sites
Soil particles have a finite number of sorption sites. As contaminant concentration increases, these sites may become saturated.
Once sorption sites approach saturation, additional contaminant molecules remain in solution rather than attaching to the soil surface.
Heterogeneous Surface Properties
Soils often contain a mixture of minerals with different sorption properties. Some surfaces may bind contaminants strongly, while others bind weakly.
This heterogeneity can produce non-linear sorption behavior.
Chemical Reactions
In some cases, sorption involves chemical reactions between contaminants and mineral surfaces.
These reactions may occur differently at different concentrations, leading to non-linear relationships.
Changes in Groundwater Chemistry
Environmental conditions such as pH, ionic strength, and redox conditions may influence sorption behavior and produce non-linear responses.
Sorption Isotherms Used in Non-Linear Modeling
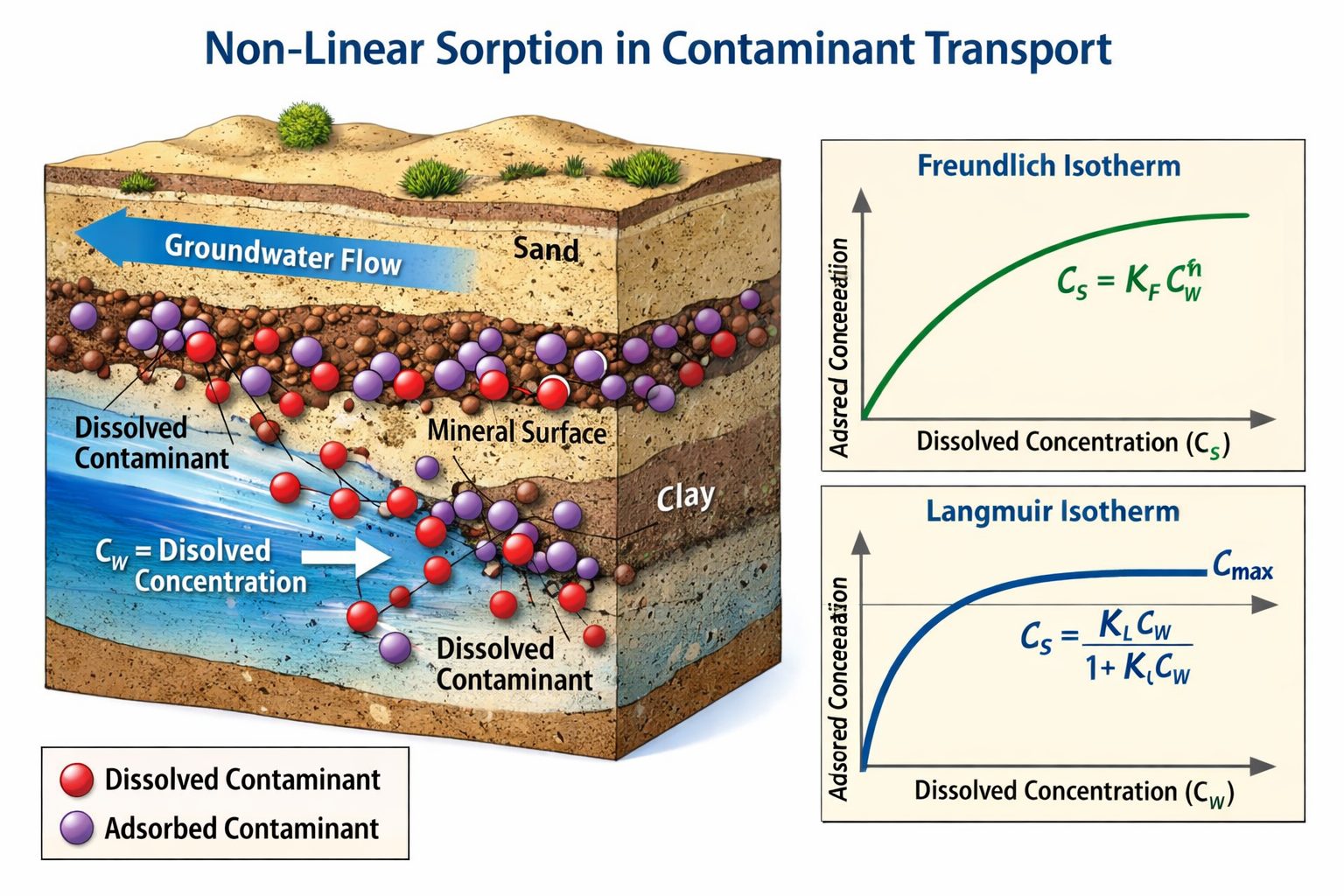
To represent non-linear sorption behavior, scientists use sorption isotherm models. These models describe how sorbed concentration varies with dissolved concentration.
Two of the most commonly used isotherm models are the Freundlich isotherm and the Langmuir isotherm.
Freundlich Sorption Isotherm
The Freundlich isotherm is an empirical model commonly used to describe non-linear sorption in environmental systems.
S = K_f C^n
Where:
- S = sorbed concentration
- C = dissolved concentration
- Kf = Freundlich sorption constant
- n = non-linearity parameter
When n = 1, the equation reduces to the linear sorption model.
When n < 1, sorption decreases as concentration increases, indicating limited sorption capacity.
The Freundlich model is widely used because it can represent a wide range of sorption behaviors observed in soils and sediments.
Langmuir Sorption Isotherm
The Langmuir isotherm describes sorption behavior when sorption sites on the soil surface become saturated.
S = \frac{S_{max} K_L C}{1 + K_L C}
Where:
- Smax = maximum sorption capacity
- KL = Langmuir sorption constant
- C = dissolved concentration
This model assumes that sorption occurs at specific sites and that each site can hold only one contaminant molecule.
The Langmuir model is commonly used when sorption processes involve strong chemical interactions with mineral surfaces.
Non-Linear Sorption in Transport Models
When non-linear sorption is included in contaminant transport models, it significantly affects contaminant plume behavior.
Unlike linear sorption, non-linear sorption causes the retardation factor to vary with concentration.
This means that contaminants may move faster or slower depending on concentration levels.
For example:
- High contaminant concentrations may saturate sorption sites, increasing mobility.
- Low concentrations may experience stronger sorption, slowing migration.
As a result, contaminant plumes may spread differently than predicted by linear models.
Impact on Contaminant Plume Migration
Non-linear sorption can produce several effects in contaminant transport modeling.
Asymmetric Plume Shapes
Non-linear sorption may cause contaminant plumes to develop asymmetric shapes because different portions of the plume experience different sorption conditions.
Concentration-Dependent Transport
High-concentration regions may move faster than low-concentration regions due to sorption site saturation.
Extended Contaminant Tails
Some contaminants may remain sorbed to soil particles and slowly release over time, producing long contaminant tails behind the main plume.
These effects are particularly important in groundwater contamination studies.
Determining Non-Linear Sorption Parameters
Accurate representation of non-linear sorption requires experimental determination of sorption parameters.
Batch Sorption Experiments
Batch experiments are commonly used to measure sorption behavior.
In these experiments:
- Soil samples are mixed with solutions containing known contaminant concentrations.
- The mixture is allowed to reach equilibrium.
- The dissolved concentration is measured.
By repeating this process at different concentrations, researchers can generate sorption isotherms.
Column Experiments
Column experiments simulate contaminant transport through soil under flowing conditions.
Contaminated water is passed through a column packed with soil, and contaminant concentrations are measured at the outlet.
Column experiments provide valuable data on sorption kinetics and transport behavior.
Field Observations
In some cases, non-linear sorption parameters may be estimated from field data using inverse modeling techniques.
Transport models are calibrated by adjusting sorption parameters until simulated contaminant plumes match observed data.
Applications of Non-Linear Sorption Modeling
Non-linear sorption modeling is used in many environmental applications.
Groundwater Contamination Studies
Accurate sorption modeling helps predict contaminant plume migration and evaluate potential impacts on drinking water resources.
Landfill Environmental Assessments
Sorption models help evaluate how contaminants from landfill leachate interact with soil and geological barriers.
Remediation Design
Non-linear sorption models can help design effective remediation strategies, including pump-and-treat systems and reactive barriers.
Agricultural Pollution Studies
Non-linear sorption modeling helps predict the transport of pesticides and fertilizers in soil and groundwater systems.
Challenges in Modeling Non-Linear Sorption
Although non-linear sorption models provide more realistic representations of contaminant behavior, they also introduce additional complexity.
Key challenges include:
Parameter Uncertainty
Sorption parameters vary widely depending on soil composition and environmental conditions.
Spatial Variability
Soil properties may vary across a site, producing different sorption behavior in different areas.
Computational Complexity
Non-linear transport equations are more complex to solve than linear models and may require advanced numerical techniques.
Because of these challenges, environmental modelers often perform sensitivity analyses to evaluate the influence of sorption parameters.
Advances in Sorption Modeling
Recent advances in environmental modeling are improving our understanding of non-linear sorption processes.
New research areas include:
- multi-site sorption models
- kinetic sorption models
- reactive transport modeling
- molecular-scale sorption studies
These approaches provide more detailed insights into contaminant behavior and improve the accuracy of environmental models.
Conclusion
Non-linear sorption plays a critical role in controlling contaminant transport in soil and groundwater systems. While linear sorption models are often used for simplicity, many contaminants exhibit complex sorption behavior that cannot be accurately represented using constant distribution coefficients.
Models such as the Freundlich and Langmuir isotherms provide more realistic representations of sorption processes and help explain how contaminants interact with geological materials.
Accurately incorporating non-linear sorption into contaminant transport models allows environmental scientists and engineers to better predict contaminant plume migration, assess environmental risks, and design effective remediation strategies.
As research continues to advance our understanding of sorption processes, improved experimental techniques and modeling approaches will further enhance our ability to simulate contaminant transport and protect groundwater resources.
Learn more about our Contaminant Transport Modeling Solutions
Related Articles
- Contaminant Transport Modelling and Landfill Design: A Complete Guide for Environmental Engineers
- How Contaminant Transport Models Predict Groundwater Pollution
- Designing Landfill Liner Systems to Prevent Groundwater Contamination
- Understanding Leachate Generation and Transport in Landfills
- Advection–Dispersion Modelling in Groundwater Systems
- Hydrogeological Data Required for Contaminant Transport Models
- Regulatory Requirements for Landfill Design and Groundwater Protection
- Unlock Global Expertise: Free Research Viewer for POLLUTE and MIGRATE
- Mastering the Plume: POLLUTEv8 vs. MODFLOW vs. FEFLOW vs. PATH3D
- Core Features of POLLUTEv8
- Mastering Contaminant Transport: Special Features of POLLUTEv8
- Navigating Contaminant Migration with POLLUTE: A Modern Approach to Landfill Design
- Beyond The MCL: Building Audit-Proof Contaminant Fate Models For 2026 Regulatory Submissions
- Using Predictive Models to Meet 2026 Primary Drinking Water Regulations
- Driving Environmental Research: How GAEA Technologies POLLUTE Software is Used in Academic and Industry Reports
- Modeling Your Way to ‘No Further Action’: How Predictive Simulation Shaves Years Off Remediation Timelines
- Meeting The New 2026 EPA Reporting Standards For PFAS: Why Traditional Spreadsheet Modeling No Longer Suffices
- Unlocking Sustainable Solutions: The Role of POLLUTE Software in Landfill Design and Contaminant Transport Modelling
- Contaminant Transport Modelling and Landfill Design Insights
- Geomembrane Degradation in Landfill Liners: Causes, Modeling, and Long-Term Performance
- Clogging of Landfill Leachate Collection Systems: Causes, Impacts, and Prevention
- Determining Diffusion Coefficients for Contaminant Transport Modeling
- Use and Determination of Distribution Coefficients for Contaminant Transport Modeling
- Phase Change in Collection Systems in Contaminant Transport Modeling for Landfills
- Biological and Radioactive Decay in Contaminant Transport Modeling
External References
- Groundwater Contaminant Migration Processes
- Soil–Water Partitioning and Sorption Processes
- Freundlich sorption isotherm for nonlinear adsorption
- Langmuir Sorption Model
- Effect of Non-Linear Sorption on Contaminant Plumes
- Non-Linear Sorption in Soil Systems
- Reactive transport modeling in porous media
- Henry’s Law and Gas–Liquid Partitioning in Landfills
- Modeling the Fate of Organic Chemicals in Landfills
- Models of Biodegradation During Contaminant Transport
- Modeling Decay Chains of Radioactive Contaminants