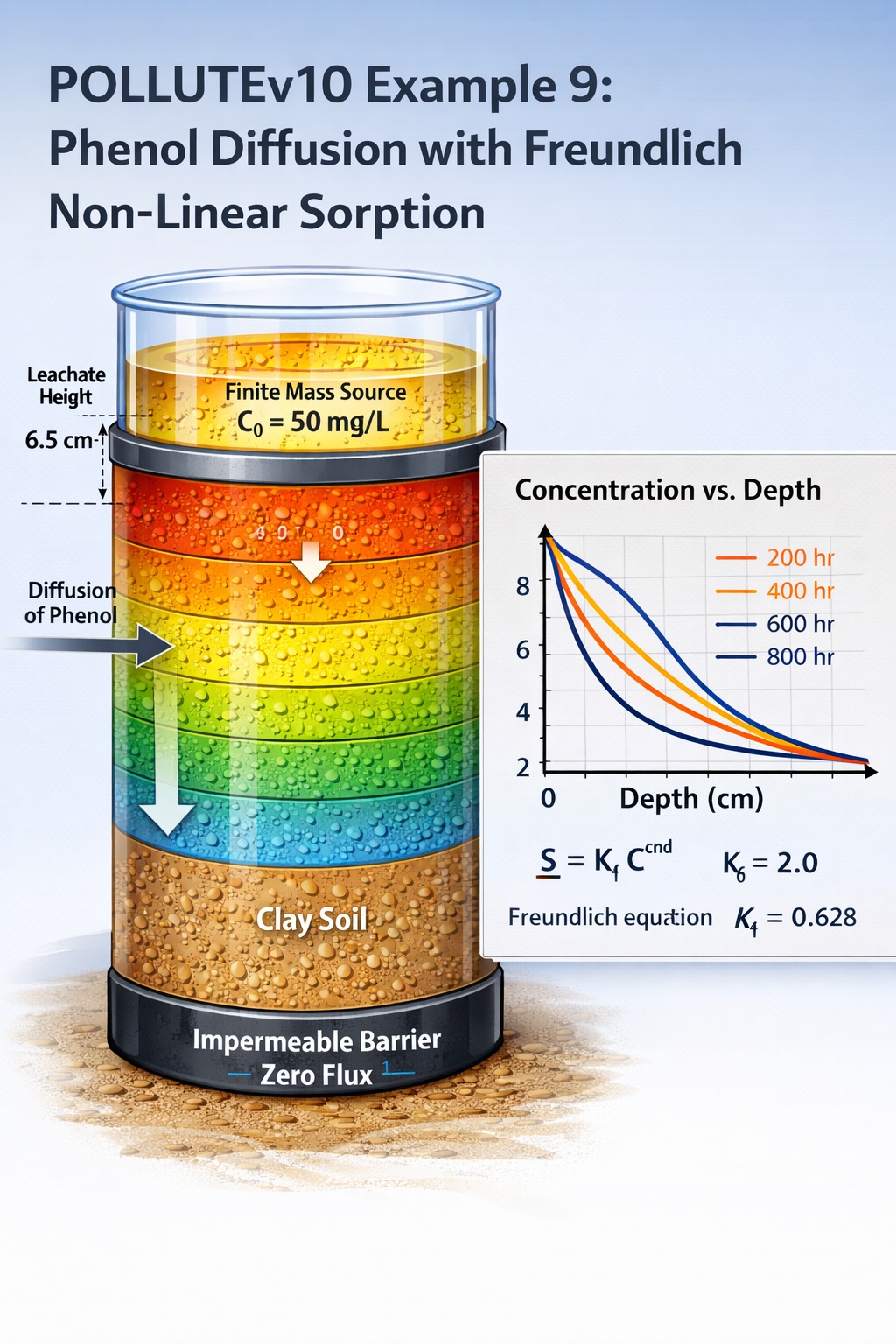
In POLLUTEv10 Example 9, the model advances beyond linear sorption by incorporating Freundlich non-linear sorption to simulate the diffusion of phenol through a clay specimen. This example reflects more realistic contaminant behavior, particularly for organic compounds that do not follow simple linear partitioning.
Problem Overview
This example simulates a laboratory diffusion test with the following conditions:
- Contaminant: Phenol
- Soil: Clay (7 cm thick)
- Transport mechanism: Diffusion only (no advection)
- Sorption model: Freundlich non-linear isotherm
- Bottom boundary: Impermeable (zero flux)
- Source type: Finite mass
Source Conditions
- Initial concentration (co): 50 mg/L
- Leachate head (Hr): 6.5 cm
Times of Interest
- 200 hr
- 400 hr
- 600 hr
- 800 hr
Conceptual Model
The system consists of:
- A finite mass source of phenol at the top
- A clay layer where diffusion and sorption occur
- An impermeable base preventing downward flux
Unlike constant concentration sources, the finite source means:
- Concentration at the top decreases over time
- Transport is influenced by both diffusion and depletion
Input Parameters
| Property | Symbol | Value | Units |
|---|---|---|---|
| Darcy Velocity | va | 0.0 | cm/hr |
| Diffusion Coefficient | D | 0.019 | cm²/hr |
| Freundlich Coefficient | Kf | 2.0 | cm³/g |
| Sorption Exponent | — | 0.628 | – |
| Soil Porosity | n | 0.46 | – |
| Dry Density | — | 1.47 | g/cm³ |
| Soil Layer Thickness | H | 7.0 | cm |
| Number of Sub-layers | — | 14 | – |
| Source Concentration | co | 50.0 | mg/L |
| Leachate Height | Hr | 6.5 | cm |
Understanding Freundlich Non-Linear Sorption
The Freundlich isotherm describes sorption as:
Where:
- S = sorbed concentration
- C = լուծ dissolved concentration
- Kf = sorption capacity
- n = non-linearity exponent
Key Implications
- Sorption is not constant (unlike linear Kd models)
- Retardation varies with concentration
- Transport becomes concentration-dependent
With n = 0.628 (< 1):
- Sorption is stronger at lower concentrations
- This causes tailing effects in concentration profiles
Key Processes Simulated
1. Diffusion
- Governed by concentration gradients
- Slower compared to Example 8 due to lower diffusion coefficient
2. Non-Linear Sorption
- Phenol interacts with clay via Freundlich behavior
- Retardation is dynamic, not constant
3. Finite Mass Source
- Source concentration decreases over time
- Results in attenuated diffusion fronts
4. Boundary Condition
- Zero flux at base → contaminant accumulates within the domain
Importance of Layer Discretization
This example highlights a critical modeling requirement:
Accuracy depends strongly on the number of sub-layers when using non-linear sorption.
Why?
- Non-linear equations require finer resolution
- Concentration-dependent retardation must be captured precisely
- Coarse discretization can lead to numerical errors
Best Practice
- Use ≥ 14 sub-layers (as in this example)
- Increase layers further for higher accuracy or steeper gradients
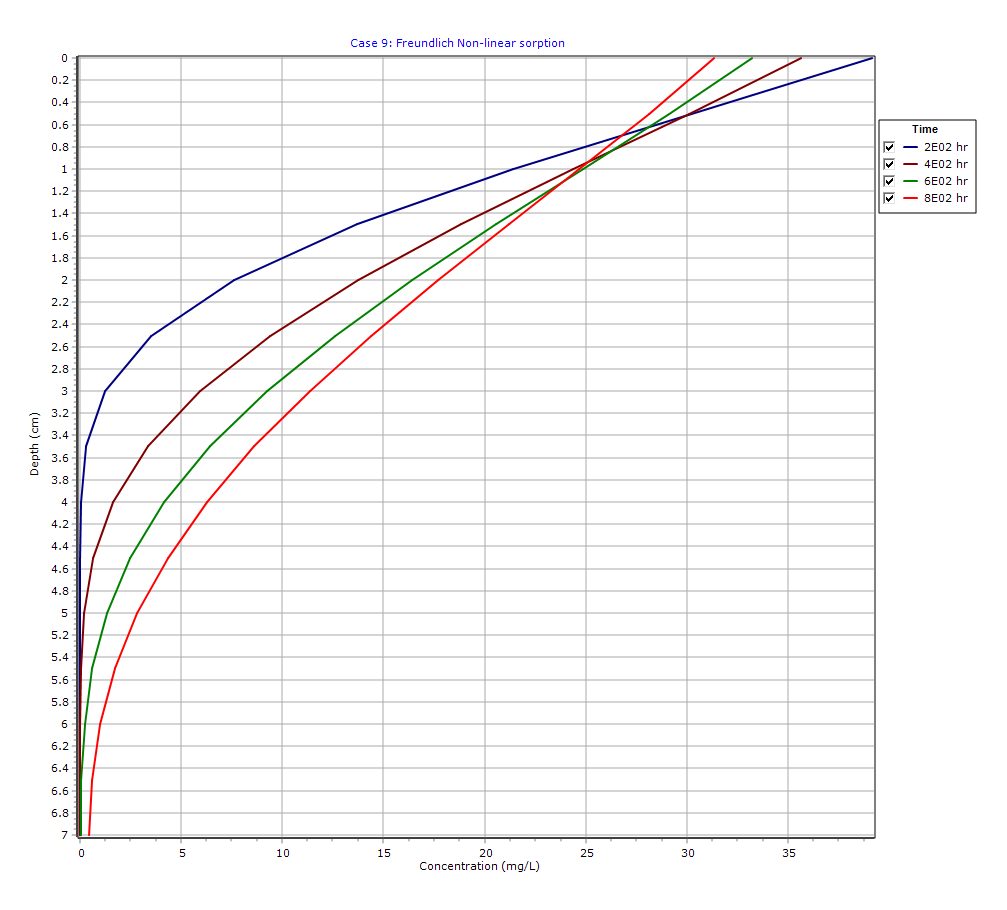
Graphical Output: Depth vs Concentration
PDF Report
 Loading…
Loading…
Concentration Profiles
- Profiles evolve over time (200 → 800 hr)
- Slower migration compared to conservative solutes
- Gradual flattening due to source depletion
Non-Linear Effects
- Stronger sorption at low concentrations causes extended tails
- Profiles are non-symmetric and more complex
Finite Source Behavior
- Peak concentrations decrease over time
- Diffusion front weakens as source mass is exhausted
Practical Applications
This example is especially relevant for:
- Organic contaminant transport (e.g., phenol, hydrocarbons)
- Landfill leachate assessments
- Clay liner performance evaluation
- Risk assessment modeling
It is particularly important when:
- Contaminants exhibit non-linear adsorption
- Long-term predictions are required
- Laboratory calibration data is available
Best Practices for POLLUTEv10 Users
- Always verify Freundlich parameters (Kf and n) from lab data
- Use fine discretization for non-linear problems
- Compare results at multiple time steps
- Be cautious when interpreting retardation (not constant!)
Conclusion
POLLUTEv10 Example 9 demonstrates how incorporating Freundlich non-linear sorption significantly enhances the realism of contaminant transport modeling.
Key takeaways:
- Non-linear sorption leads to concentration-dependent transport
- Finite sources introduce time-varying boundary conditions
- Numerical accuracy depends heavily on layer discretization
This example is essential for modeling organic contaminants in clay systems, where linear assumptions are often insufficient.
Learn more about our Contaminant Transport Modeling Solutions
POLLUTE Examples
- POLLUTEv10 Example 1: Modeling a U.S. RCRA Subtitle D Landfill
- POLLUTEv10 Example 2: Pure Diffusion in a Soil Layer (No Sorption)
- POLLUTEv10 Example 3: Advection + Diffusion with Aquifer Mixing
- POLLUTEv10 Example 4: Finite Mass Source with Leachate Collection System
- POLLUTEv10 Example 5: Hydraulic Trap (Upward Flow into the Landfill)
- POLLUTEv10 Example 6: Fractured Layer with Sorption and Reactive Transport
- POLLUTEv10 Example 7: Lateral Migration of a Radioactive Contaminant in Fractured Rock
- POLLUTEv10 Example 8: Laboratory Diffusion of Potassium in Clay
- POLLUTEv10 Example 10: Time-Varying Advective–Dispersive Transport from a Landfill
- POLLUTEv10 Example 11: Time-Varying Source Concentration with Diffusion (Chloride in Clay)
- POLLUTEv10 Example 12: Fractured Media Transport vs Analytical Solution (Tang et al., 1981)
- POLLUTEv10 Example 13: 2D Plane Dispersion vs Analytical Solution (TDAST)
- POLLUTEv10 Example 14: Modeling a Landfill with Primary and Secondary Leachate Collection Using Passive Sink
- POLLUTEv10 Example 15: Modeling Leachate Collection System Failure Using Variable Properties and Passive Sink
- POLLUTEv10 Example 16: Monte Carlo Simulation of Leachate Collection System Failure Timing
- POLLUTEv10 Example 17: Modeling a Landfill with Composite Liners and Dual Leachate Collection Systems
- POLLUTEv10 Example 18: Modeling Phase Change in a Secondary Leachate Collection System
- POLLUTEv10 Example 19: Multiphase Diffusion of Toluene Through a Geomembrane System
- POLLUTEv10 Example 20: Sensitivity Analysis of Primary Leachate Collection System Failure
Comparison between POLLUTE and MIGRATE
- MIGRATEv10 vs POLLUTEv10: Pure Diffusion Comparison
- MIGRATEv10 vs POLLUTEv10: Advective–Diffusive Transport Comparison
- MIGRATEv10 vs POLLUTEv10: Finite Mass Source Comparison
- MIGRATEv10 vs POLLUTEv10: Hydraulic Trap (Finite Mass Source) Comparison
- MIGRATEv10 vs POLLUTEv10: Fractured Layer with Sorption Comparison